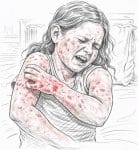
Severe atopic dermatitis represents the most debilitating end of the spectrum of one of the most prevalent chronic inflammatory skin diseases in the world, producing a degree of physical suffering, functional impairment, psychological distress, and quality of life disruption that in rigorous health utility studies is comparable to or exceeds that of severe heart failure, major depressive disorder, and end-stage renal disease. This ranking of severe atopic dermatitis among the most impactful of all chronic diseases by patient-reported outcome measures reflects the comprehensive, relentless, and multi-dimensional nature of its burden: the constant, often unbearable pruritus that dominates waking hours and disrupts sleep to the point of producing a state of chronic sleep deprivation; the widespread skin inflammation, lichenification, and weeping that make physical appearance a source of shame and social avoidance; the perpetual risk of infection and the uncertainty of unpredictable flares; and the psychological consequences of living with a highly visible, stigmatizing condition that profoundly affects the way others respond to the affected individual in personal and professional contexts.
The evolution of atopic dermatitis treatment over the past decade has been remarkable, driven by an explosion in the understanding of the molecular immunology of the condition that has identified specific therapeutic targets amenable to blockade by precisely engineered biological agents and small molecule kinase inhibitors. Prior to 2017, the treatment options for severe atopic dermatitis that had failed topical therapies were limited to traditional immunosuppressive agents including cyclosporine, azathioprine, and methotrexate, each carrying substantial risks of serious adverse effects that limited their long-term use and rendered them unsuitable for many patients including children. The approval of dupilumab in 2017, followed by the subsequent approvals of additional biological agents and oral JAK inhibitors, has transformed the treatment landscape for severe atopic dermatitis, providing for the first time a generation of therapies that can achieve near-complete or complete skin clearance in a substantial proportion of previously treatment-refractory patients without the systemic immunosuppression that characterized the older treatment options.
The clinical definition of severe atopic dermatitis encompasses the quantitative assessment of skin involvement and symptom burden using validated severity measures alongside the clinical judgment of experienced dermatologists. The Eczema Area and Severity Index, the SCORing Atopic Dermatitis instrument, the Investigator Global Assessment, and the Patient Oriented Eczema Measure each provide validated quantitative assessments of different dimensions of atopic dermatitis severity, with the Eczema Area and Severity Index score above twenty-three and the SCORing Atopic Dermatitis score above fifty conventionally defining severe disease in clinical research settings. The incorporation of patient-reported symptom burden measures including the weekly average peak pruritus numerical rating scale and the Dermatology Life Quality Index alongside these physician-assessed measures provides a more complete picture of disease impact that encompasses both the objective skin disease and its subjective consequences for the patient’s daily experience.
Immunological Mechanisms of Severe Atopic Dermatitis
The immunological complexity of severe atopic dermatitis has been progressively elucidated by molecular profiling studies that have characterized the cytokine and cellular landscape of lesional and non-lesional atopic skin at transcriptomic, proteomic, and single-cell resolution, revealing a disease that is driven by multiple interconnected inflammatory pathways rather than the simple Th2-dominated model that dominated early atopic dermatitis immunology. While Th2-mediated inflammation driven by interleukin-4 and interleukin-13 remains the central immunological axis of atopic dermatitis, the severe end of the spectrum is characterized by the activation of additional inflammatory pathways including the Th1, Th17, and Th22 axes that layer additional cytokine signals onto the Th2 substrate and contribute to the distinctive features of severe chronic atopic dermatitis including profound lichenification and the Th17-mediated barrier disruption that is particularly prominent in Asian atopic dermatitis phenotypes.
Interleukin-4 and interleukin-13, which signal through a shared receptor complex incorporating the interleukin-4 receptor alpha chain, are the master regulators of the atopic inflammatory response and drive the disease through multiple simultaneous mechanisms. Both cytokines suppress the expression of the genes encoding filaggrin, loricrin, and involucrin in keratinocytes, reducing the production of the structural proteins that maintain stratum corneum integrity and producing the skin barrier dysfunction that is both a cause and a consequence of the inflammatory cascade. Interleukin-4 drives the differentiation of naive T helper cells toward the Th2 phenotype and promotes the class switching of B lymphocytes to produce immunoglobulin E, the antibody class whose elevated production characterizes the atopic state and whose binding to high-affinity receptors on mast cells and basophils primes these cells for rapid allergen-triggered degranulation. Interleukin-13 drives goblet cell-like metaplasia of epidermal keratinocytes with increased mucin production, promotes dendritic epidermal hyperplasia, and directly activates sensory neurons through the IL-13 receptor alpha 2 expressed on dorsal root ganglion neurons, contributing to the itch signaling that drives the scratch-itch cycle perpetuating skin damage.
Interleukin-31, produced predominantly by Th2 lymphocytes and elevated in the skin and blood of severe atopic dermatitis patients in proportion to disease severity and itch intensity, acts on a receptor expressed on cutaneous sensory neurons including the C-fiber nociceptors responsible for itch transduction, producing the intense, burning, and refractory pruritus that patients with severe atopic dermatitis consistently identify as the most disabling feature of their disease. The direct neuronal targets of interleukin-31 explain why the itch of severe atopic dermatitis responds poorly to conventional antihistamines that block only histaminergic itch pathways and why it requires agents targeting the interleukin-31 pathway, including the anti-interleukin-31 receptor alpha monoclonal antibody nemolizumab, for meaningful relief of this specific symptom dimension. Thymic stromal lymphopoietin, an epithelial cytokine released by keratinocytes in response to barrier disruption, environmental allergens, proteases, and mechanical trauma from scratching, serves as a crucial upstream initiator of the atopic inflammatory cascade by activating dendritic cells, mast cells, basophils, and innate lymphoid cells type 2 to produce the Th2-polarizing cytokine environment that drives atopic dermatitis pathogenesis.
Burden of Disease and Psychosocial Impact
The burden of severe atopic dermatitis extends far beyond the skin to encompass the psychological, social, occupational, and economic dimensions of a chronic, visible, and stigmatizing condition that interferes with virtually every aspect of the affected individual’s life. Sleep disruption, driven by the intense nocturnal pruritus that characterizes atopic dermatitis and that is mediated in part by the normal nocturnal rise in skin temperature that lowers the threshold for itch perception, is one of the most universally reported and most objectively measurable impacts of severe atopic dermatitis, with polysomnographic studies demonstrating significantly reduced sleep efficiency, increased number of awakenings, and reduced slow-wave sleep in severe atopic dermatitis patients compared to healthy controls. The chronic sleep deprivation resulting from nightly itch-induced disruption produces the cognitive impairment, emotional dysregulation, and physical fatigue that impair school and work performance and contribute to the depression and anxiety that are highly prevalent comorbidities in severe atopic dermatitis.
Depression affects approximately thirty to forty percent of adults with severe atopic dermatitis, a rate two to three times higher than in the general population, reflecting both the neurobiological consequences of chronic pain and pruritus on mood regulation and the psychological impact of living with a highly visible, socially stigmatizing condition that affects self-image, intimate relationships, and public social functioning. The shame and embarrassment associated with visibly inflamed, weeping, and excoriated skin in public settings drives the social withdrawal and avoidance behaviors that compound the direct functional limitations of the disease, progressively constricting the affected individual’s world to settings in which they feel safe from the stares and questions of those unfamiliar with their condition. Work and school absenteeism attributable to severe atopic dermatitis is substantial, with affected individuals reporting significantly more days of missed work or school per year than matched controls, contributing to both individual economic disadvantage and broader workforce productivity losses.
The economic burden of severe atopic dermatitis encompasses the direct costs of medical care including dermatology consultations, hospitalizations for severe flares and complications, prescription medications, and over-the-counter skincare products alongside the indirect costs of lost productivity from absenteeism, presenteeism, and caregiver time devoted to assisting affected children or adults. The introduction of biological therapies for severe atopic dermatitis has added a new dimension of direct treatment cost, with dupilumab and the subsequently approved biological agents carrying annual treatment costs of twenty to forty thousand US dollars per patient that have generated discussions about cost-effectiveness and healthcare resource allocation. The health economic analyses that have evaluated the cost-effectiveness of biological therapies for severe atopic dermatitis generally find favorable cost-effectiveness ratios when the full burden of severe disease, including indirect costs, quality-adjusted life years, and complication prevention, is incorporated into the analysis.
Biological and Targeted Therapies
The therapeutic revolution in severe atopic dermatitis that began with the approval of dupilumab in 2017 has continued with the approval of additional biological agents and oral targeted therapies that collectively provide multiple distinct therapeutic options for patients with inadequately controlled severe atopic dermatitis. Dupilumab, a fully human monoclonal antibody targeting the interleukin-4 receptor alpha chain that blocks the shared signaling receptor of both interleukin-4 and interleukin-13, has established itself as the foundational biological therapy for severe atopic dermatitis, with Phase 3 clinical trial data demonstrating that approximately forty percent of patients achieve a seventy-five percent reduction in the Eczema Area and Severity Index score and that approximately thirty percent achieve a ninety percent reduction, representing near-complete or complete skin clearance in a substantial proportion of previously treatment-refractory patients. The favorable long-term safety profile of dupilumab, reflecting its selective targeting of the Th2 cytokine axis without broad immunosuppression, enables its use for prolonged durations including in pediatric patients from six months of age.
Tralokinumab, an anti-interleukin-13 monoclonal antibody that specifically targets interleukin-13 without affecting interleukin-4 signaling, and lebrikizumab, another anti-interleukin-13 antibody with a distinct binding epitope, provide alternative biological options with similar efficacy profiles to dupilumab and the possibility of superior tolerability in patients who experience conjunctivitis, the most common treatment-emergent adverse effect of dupilumab attributed to its simultaneous blockade of interleukin-4-mediated ocular surface protection alongside interleukin-13. These interleukin-13-selective agents have demonstrated significant improvements in skin clearance, pruritus, and quality of life in Phase 3 trials of severe atopic dermatitis patients, establishing the interleukin-13 pathway as the dominant therapeutic target among the biological therapies currently available for the condition.
Oral JAK inhibitors targeting the Janus kinase enzymes that mediate intracellular signaling downstream of multiple cytokine receptors involved in atopic dermatitis pathogenesis, including upadacitinib targeting JAK1 and abrocitinib also targeting JAK1, have demonstrated rapid and dramatic improvements in severe atopic dermatitis in Phase 3 clinical programs, with head-to-head comparisons against dupilumab demonstrating superior pruritus relief and comparable or superior skin clearance at the higher doses studied. The rapid onset of action of oral JAK inhibitors, with meaningful itch reduction often apparent within twenty-four to forty-eight hours of initiating treatment, compared to the four to eight week timeframe for full dupilumab effect, represents a clinically important advantage for patients with severe active disease whose most urgent treatment need is rapid relief of disabling pruritus. The safety profile of oral JAK inhibitors requires specific monitoring for the adverse effects identified across the JAK inhibitor class including increased risk of serious infections, herpes zoster reactivation, and the cardiovascular and thromboembolic risks that led to class-wide labeling updates, though the atopic dermatitis populations studied appear to have lower background cardiovascular risk than the rheumatoid arthritis populations in which the most significant safety signals were identified.